/cdn.vox-cdn.com/uploads/chorus_image/image/73162831/2036911067.0.jpg)
Highlights! Brian Ortega badly rolls ankle during intros, pulls off gutsy submission over Yair Rodriguez | UFC Mexico City - MMAmania.com

Santhera and ReveraGen Complete NDA Submission to FDA for Vamorolone in Duchenne Muscular Dystrophy - CureDuchenne
Legend Biotech, J&J finish off rolling submission for CAR-T hopeful, approach finish line | Fierce Biotech

SpringWorks on X: "We are happy to have initiated the rolling submission of our New Drug Application with the FDA for our investigational MEK inhibitor in pediatric and adult patients with neurofibromatosis

FALA- Friedreich Ataxia Lebanese Association - ---- REPOST ---- FARA تقدم رائع! لكل المهتمين ببعض التوضيحات حول الأومافيلوكسولون: - أعلنت شركة الأدوية REATA أنه تم تقديم جزءًا من طلب الدواء الجديد (NDA)





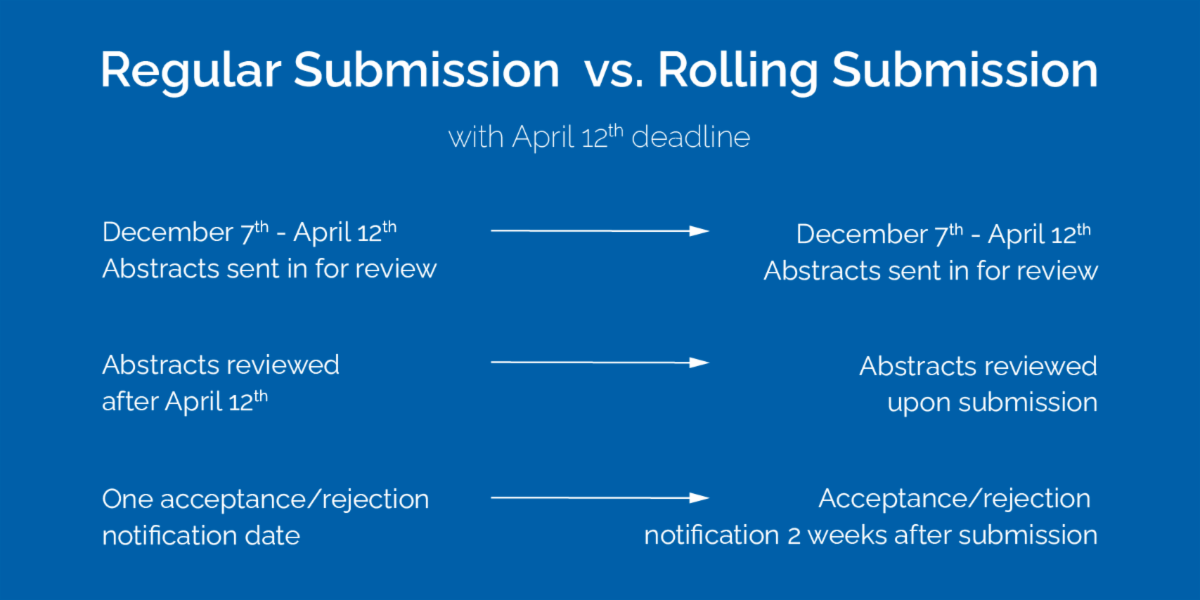



![Call for Blogs | Law and Pop-Culture Forum | MNLU Mumbai [Rolling Submission] | SCC Times Call for Blogs | Law and Pop-Culture Forum | MNLU Mumbai [Rolling Submission] | SCC Times](https://www.scconline.com/blog/wp-content/uploads/2023/10/Law-and-Pop-Culture-Forum.webp)









